Scientific journal
European Journal of Natural History
ISSN 2073-4972
ИФ РИНЦ = 0.204
ADАPTIVE TRANSFER OF THE IMMUNE RESPONSE – NEW APPROACHES
The urgency of the problem. Adaptive transfer (AT) is known to be the transfer of an immune response to a specific antigen from an immunized donor to an intact recipient. There are several variants of adaptive transfer realization. The most commonly used transfer of immunocompetent cells (lymphocytes) between monozygous twins or syngeneic animals, one of which is sensitized to a specific antigen [1]. Currently, adaptive transfer is attracting attention due to the neoplastic diseases treating possibility [2, 3]. Adaptive cell therapy (ACT) of cancer patients is gaining more and more importance in practical medicine [4, 5]. ACT is carried out using some subpopulations of T lymphocytes – for example, CCR7+, CD27+, CD28+, CD62L+ phenotypes [6, 7, 8], natural killer cells (NK cells), macrophages [9].
The aim of this investigation was to study the possibility of platelets using for adaptive transfer of the primary immune response of BALB/c mice to sheep red blood cells (SRBC) to syngeneic recipients.
Materials and research methods
The male BALB/c mice weighing no more than 20.0 grams were used performing this work. The mice were kindly provided by the nursery of the Technological Center of the Academy of Sciences of Turkmenistan. Mice of this strain are known to be highly responsive to SRBC immunization and are widely used in adaptive transfer studies [10]. Two series of experiments were performed. The design of study is shown in the figure (Fig. 1). In the first series of experiments, 15 mice were used, which were injected intraperitoneally with 0.1 ml of a 20 % suspension of SRBC. The immunized animals were examined before -, on days 3, 7, 14 and 21 after immunization.
The expression of the primary immune response of animals to SRBC was judged by the character of changes in the cellular composition of peripheral blood, the value of the granulocyte index (GI), the number of rosette-forming cells in the spleen (RFCs), the value of the leukocyte migration index (LMI) in the presence of SRBC lysate in a modified reactions of leukocytes migration inhibition [11].
At the peak of primary immune response to SRBC (days 7-8), some mice were used as donors of spleen lymphocytes (SL) and platelets (PLT).
The animals were removed from the experiment under ether anesthesia by dislocation of the cervical vertebrae, the spleen was removed, homogenized on an ice substrate using a Potter homogenizer, washed with saline sodium chloride solution, and a lymphocyte suspension was prepared on it at a rate of 2x106/ml. At the same time, platelets (PLT) were isolated from the peripheral blood of syngeneic donors at the peak of the immune response to SRBC by the method of two-stage centrifugation [12], and a suspension was prepared at the rate of 2x106 / ml saline sodium chloride.
In the second series of experiments, 2 groups of animals, 10 mice in each, were identified – they served as recipients of drugs and PLT from syngeneic donors immunized with SRBC. The recipients of the 1st group were injected intraperitoneally with 0.1 ml of the spleen lymphocyte suspension; the recipients of the 2nd group were injected with 0.1 ml of the PLT suspension. On days 3, 7, 14 and 21 after immunization, the recipient animals of both groups were examined according to the same scheme as in the first series (see the study design in Fig. 1). The data obtained were mathematically processed using the SPSS program (USA).
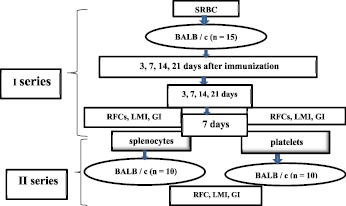
Fig. 1. Study design
A (SRBC) B (SL)
C (PLT)
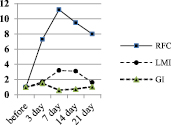
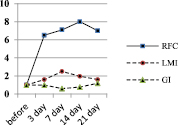
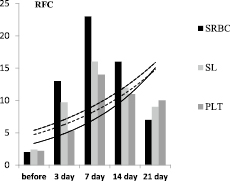
Fig. 2. Dynamics of the characteristics of the immune response of BALB/c mice to SRBC – A, spleen lymphocytes (SL) – B, and platelets (PLT) – C
Research results and discussion
In the first series of experiments, the primary immune response of BALB/c mice to SRBC was expressed by the accumulation of RFC in the spleen, an increase in the LMI value, and characteristic changes in the GI value. The dynamics of the degree of changes in these characteristics of the immune response in relation to the initial level, taken as 1, is shown in Fig. 2.
The diagram shows that the immune response of mice to SRBC (Fig. 2A) is maximally expressed on the 7th day from the moment of immunization. At this time, the number of RFC in the spleen is maximum and is 23.6 ± 1.3×103 per 106 lymphocytes versus 2.3 ± 0.01×103 per 106 before immunization – that is, it increases by 11 times against the initial level. Then, in the dynamics of the immune response, the LMI gradually increases and on the 3rd day it significantly differs from the initial level (p < 0.05), becomes the maximum on the 7th day, being at 187.6 ± 11.2 versus 59.7 ± 1.9 cu before immunization. That is, by the 7th day, the LMI increases 3.2 times. Starting from the 14th day, the LMI gradually decreases, but on the 21st day it remains significantly higher than the initial level (p < 0.05).
After immunization of mice with SRBC, the GI value increases rapidly during the first three days and is 1.17 ± 0.03 a.u. against 0.72 ± 0.05 c.u. before immunization (p < 0.01). By the 7th day, the GI significantly decreases – to 0.5 ± 0.03 (the difference is mathematically significant both in relation to 7 days and the baseline, p < 0.05 in both cases). By the 21st day, the GI practically reaches the initial level (0.8 ± 0.07 c.u., p > 0.05).
In the second series of experiments, in response to the administration of spleen leucocytes (SL) to recipients from syngeneic donors previously immunized with SRBC, the number of RFC in the spleen also increases (Fig. 2 C), but at a later date – by 14 days and to a lesser extent in comparison with the primary immunized animals – 8 times. At the same time (on the 14th day), the LMI in recipients increases maximally, but 3.2 times in relation to the initial level. The GI value, in contrast to the animals immunized with SRBC, significantly decreases only on the 7th day, then it progressively increases and by the 21st day it is 1.2 ± 0.03 (the difference is significant in relation to the initial level, p < 0.05). When the recipients are injected with a suspension SL obtained from syngeneic donors sensitized to SRBC, a changes in indicators is observed, corresponding to those in animals immunized with SRBC suspension. The responce in this case develops somewhat more slowly and it is less pronounced, but it is available.
Intraperitoneal administration of syngeneic PLT from donors previously immunized with SRBC to recipients also showed an increase in the number of RFC in the teardrop, an increase in the LMI value and modulation of GI values, which are characteristic of the immune response to only SRBC administration (Fig. 2 C). Namely – on the 7th day after the administration of PLT, the number of RFC in the spleen was 11.7 ± 5.2 x 103 per 106 SL, which is significantly lower than during immunization of animals with SRBC. However, the difference is significant in relation to the group of mice immunized with SRBC suspension (p < 0.05) and not significant in relation to the group of recipients of syngeneic SL from immune donors (p > 0.05). At the same time, the LMI value in the group of PLT recipients has a dynamics similar to that in the mice of the previous group, which received syngeneic drugs. The LMI value in this case is 132.6 ± 11.3 % versus 150.3 ± 9.8 in the group of SL recipients, that is, slightly lower compared to SL recipients (p > 0.05), but 1.9 times above the level of intact control – non-immunized animals (p < 0.01) (Fig. 2 C).
The most informative criterion for assessing the severity of the mice immune response expression to SRBC is the number of antibody-forming and rosette-forming cells in the spleen of an immunized animal. However, we found that the LMI value in the presence of SRBC lysate is an equally informative, accessible and life-saving test throughout the experiment [13].
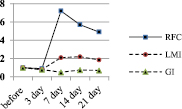
The absolute values of LMI and the number of RFC in the spleen depending on the inducer of the immune response are shown in Fig. 3 (A and B).
The diagram clearly shows that in all cases, but to varying degrees in response to immunization, there is a gradual increase in the number of RFC in the spleen and the value of LMI. Along with this, the GI value modulates in a peculiar way. If the number of RFC and the value of LMI in the dynamics of the immune response gradually increase by 7 days, then the value of GI sharply increases during the first 3 days and then by 21 days it progressively decreases to the control level. Thus, the formation of an immune response to an antigen is preceded by a sharp release of granulocytes into the bloodstream, that is, an inflammatory response from the immunized organism develops. The rate of the inflammatory response development clearly depends on the inducer of the immune response. It is maximal upon mice immunization with SRBC and minimal upon administration of PLT. But in any case, it is developing.
We have previously shown that an increase in the number of the RFC and an increase in LMI in mice immunized with SRBC correlate with hemagglutinin titers, GI value and indicate the formation of a primary immune response to this antigen [11]. With the introduction of syngeneic SL from immune donors to recipients, the same changes in indicators are observed as with the introduction of SRBC. This allows us to speak about the transfer of the immune response to SRBC from sensitized donors to syngeneic recipients. That is, an adaptive transfer of the immune response to SRBC to intact syngeneic recipients was carried out via SL.
 А
А 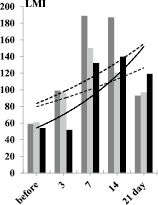 B
B
Fig. 3. Dynamics of changes in the number of RFC in the spleen of mice (in 103 per 106 ml) (A) and the absolute value of LMI, depending on the inducer of the immune response
When a PLT suspension from syngeneic donors sensitized to SRBC was administered to mice, an increase in the number of RFC in the spleen and a significant increase in LMI values were also observed in recipients. The trend lines in the charts (Fig. 3) are identical. That is, recipients of syngeneic SL and PLT respond to the SRBC antigen in vitro and in vivo in the same way as mice immunized with SRBC only, but to a somewhat lesser extent and at a later date. In other words, the results of the study suggest the possibility of adaptive transfer of sensitization to thymus-dependent corpuscular antigen – SRBC not only with the help of SL, but also platelets of the immune syngeneic donor.
Until recently, platelets were not considered blood cells, but were considered pieces of cytoplasm that are detached from megakaryocytes and enter the bloodstream exclusively as components of hemostasis [14, 15]. This common misconception was based on the fact that platelets have no nucleus. In the last 2-3 decades, platelets have increasingly attracted the attention of researchers, not only as participants in hemostasis processes, but also as participants in allergic and immunological reactions of the body [16, 17, 18]. As it turned out, platelets are rather complexly organized. They are able to regulate the expression of their own gene, synthesize protein de novo, and release various mediators with paracrine effects that affect the function of other cells in the body [19]. Platelets have complex post-transcriptional mechanisms, which allow them to change their proteome, phenotype and function, performing active protein synthesis in response to cellular activation [20, 21, 22]. Platelets are the first to initiate antimicrobial protection by detecting the presence of pathogens or products of inflammation through multiple immune receptors [23, 24, 25, 26] and primarily Toll-like receptors (TLR) [27]. Many cytokines secreted by platelets have direct antimicrobial properties [28]. In the other words, there is a lot of convincing evidence of platelet immune competence.
If the existence of functionally different leukocytes subpopulations is proven and generally accepted, then this cannot be said about platelets. The existence of platelet subpopulations has been proven relatively recently [29]. As a result of studying the biochemical interactions of blood coagulation proteins and platelets, platelet subpopulations were discovered that sharply differ in their participation in membrane and aggregation processes, information was obtained on the physiological and clinical significance of platelet division into subpopulations [30, 31]. Platelet subpopulations are differentiated according to different criteria: cell size, its response to activation by thrombin [32, 33, 34] and other humoral factors, for example adenosine 5’diphosphate [35, 36]. Of great interest is a subpopulation of the so-called “coated platelets” [37, 38, 39, 40]. In hemorrhagic syndrome, a subpopulation of “gray platelets” – abnormal platelets characterized by the absence of α-granules – was revealed. They are larger in size, have a gray color, but do not have a granulomere and not capable of aggregation [41, 42, 43, 44].
A powerful receptor apparatus, a huge number of organelles, the ability to neosynthetic processes, a significant amount of RNA, including mRNA, micro-RNA, ribosomal and transport RNA and non-coding RNA [45, 46, 47, 48], determine many functions of platelets, including immune ones.
Adaptive cell therapy stays an important problem of modern [49]. But, it is well known, that adaptive transfer of the immune response to the any antigen is possible only upon transplantation of syngeneic antigen-sensitized immunocompetent cells to the donor. The structure of platelets, their functional and morphological features, active participation in immune and inflammatory reactions, the ability to control the reactions of the immune system, allow, in our opinion, to assume the existence of an immunocompetent subpopulation of platelets. Apparently, there is a certain subpopulation of platelets capable of storing the memory of the antigen introduced into the body and transmitting information about it to the recipient and not only syngeneic.
Conclusion
The study made it possible to reveal the ability of platelets from immunized donors – BALB/c mice, for example – to transfer the state of sensitization to a specific antigen to a syngeneic recipient. That is, to carry out in the syngeneic system the adaptive transfer of the immune response to a specific antigen – in this case, a thymusdependent corpuscular antigen, which is SRBC? In this regard, we consider it promising to study the possibility of using platelets for the implementation of an adaptive transfer of the immune response to intact recipients in the allogeneic system, as well as to investigate the possibility of using platelets in adaptive cell therapy.
Further research in this direction, in our opinion, is of both theoretical and practical interest and will be continued by us.
Библиографическая ссылка
Pleskanovskaya S.A. ADАPTIVE TRANSFER OF THE IMMUNE RESPONSE – NEW APPROACHES // European Journal of Natural History. 2020. № 6. ;URL: https://world-science.ru/en/article/view?id=34134 (дата обращения: 28.04.2026).
DOI: https://doi.org/10.17513/ejnh.34134

